Discussion Forum : P-block Group 15 - Pnictogens
Question -
HNO2 (Nitrous acid) is a monobasic acid. – True or False?
Answer: Option A
:
A
HNO2 (Nitrous acid) is monobasic acid because it can donate only a single proton.
Basicity is the number of protons that an acid can give. From the structure,
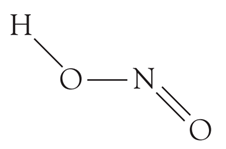
We can see that there is only one H atom and so only one proton can be abstracted from a HNO2 molecule. What can you say about the basicity of nitric acid?
Was this answer helpful ?
:
A
HNO2 (Nitrous acid) is monobasic acid because it can donate only a single proton.
Basicity is the number of protons that an acid can give. From the structure,

We can see that there is only one H atom and so only one proton can be abstracted from a HNO2 molecule. What can you say about the basicity of nitric acid?
Was this answer helpful ?
Submit Your Solution hear: