Kinetic Theory Of Gases(11th And 12th > Physics ) Questions and Answers
Explanation:-
Answer: Option D. -> TP1≠TP2.:
D
Just like a single macroscopic pressure being undefined for a gas which is not in equilibrium, a single macroscopic temperature is also undefined. So the temperature at P1 and P2 need not necessarily be the same, and most likely wouldn't be the same.
Explanation:-
Answer: Option C. ->
:
C
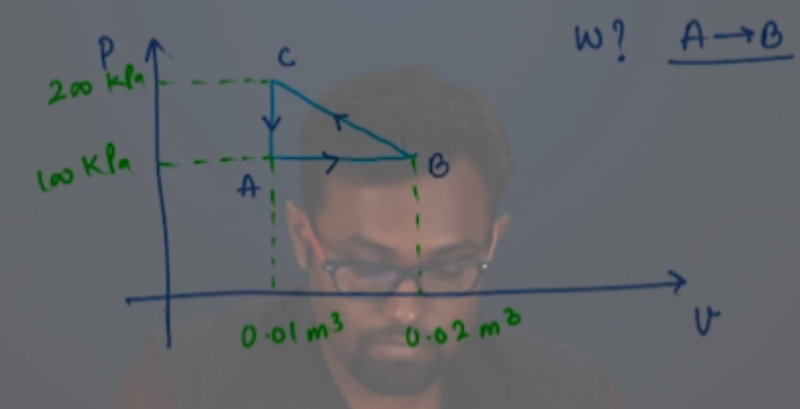
Let us analyse each process separately. BC is an isothermal process, which implies that the P-C graph would be represented by a rectangular hyperbola. (Since PV = constant).
This rectangular hyperbola starts at a lower volume V1 and goes to a higher volume as represented in the figure below -
Process AB is an isochoric process, which in a P-V graph would be a vertical line. Also we notice that the temperature drops in this process, whichimplies that the pressure will also drop (since PV=nRT, when V is constant P∝T). Therefore point Acan be anywhere on the line represented in blue in the figure below.
This process is an isobaric process since it represents a curve where V∝T (this is possible only when pressure is a constant, remember Charles' law?). So the process can be represented by a horizontal line in a P-Vgraph. The point where the line joining process A-B and C-A meet is the point A.
The Bat Life: Batman is undercover, and to prevent anyone from recognizing his voice (or Bruce Wayne's) he inhales a very dilute Helium gas (search YouTube for "voice after helium” to see the effects). The helium is stored ina cylindrical chamber as shown.
During a fight one of the steel balls gets dislodged. What will happen to the piston?
Explanation:-
Answer: Option B. -> It will move up:
B
Initial free body diagram -
When the system was in equilibrium, the force applied by the steel balls on the piston was offset by the force applied by the gas on the piston.
Just after removing the ball -
The instant the ball is dislodged the downward force acting on the piston reduces from 19.6 N (i.e., 2×9.8 N) to 14.7 N (1.5× 9.8 N). However the pressure of the helium gas has not changed. Hence there is an unbalanced force in the upward direction. This upward force will push the piston up. After some time, the weight of the balls would equal the force exerted by the helium gas and the system would eventually come to rest.
Explanation:-
Answer: Option B. -> Eint always decreases as temperature decreases:
B and D
Eint is defined as
Eint=(32)kT×N
Eint is always going to increase when temperature is going to rise. The temperature of any system is the average kinetic energy of the atoms in the system.
Explanation:-
Answer: Option B. -> Open system:
B
Let's talk about the different kinds of system first.
Open system -matter and energy both can be transferred.
Closed system - Only energy can be transferred.
Isolated - Neither energy nor matter can be transferred.
A mug is a vessel, it allows for the exchange of matter and energy between the system and surroundings. Thus it is an open system.
Explanation:-
Answer: Option B. -> False:
B
A system which exchanges both matter & energy with the surroundings is called an open system by definition.
Open system -matter and energy both can be transferred.
Closed system - Only energy can be transferred.
Isolated - Neither energy nor matter can be transferred.
Explanation:-
Answer: Option A. -> True:
A
Fundamental properties which determine the state of a system are referred to as state variables or state functions or thermodynamic parameters. The change in the state properties depends only on the initial and final states of the system, but is independent of the manner in which the change has been brought about. In other words, the state properties do ont depend upon the path taken.