Biomolecules(12th Grade > Biology ) Questions and Answers
Question 2. Match the biological molecules listed under column I with their biological functions listed under column II.
Choose the answer which gives correct combination of alphabets of two columns.
Biological MoleculeFunctiona. Glycogenp. Hormoneb. Globulinq. Biocatalystc. Steroidsr. Antibodyd. Thrombins. Storage product
Choose the answer which gives correct combination of alphabets of two columns.
Biological MoleculeFunctiona. Glycogenp. Hormoneb. Globulinq. Biocatalystc. Steroidsr. Antibodyd. Thrombins. Storage product
Explanation:-
Answer: Option A. -> a—s, b—r, c—p, d—q:
A
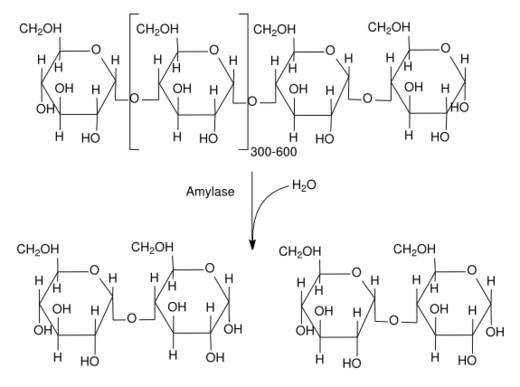
Glycogen is a polysaccharide that is stored as reserve food material in animals. Globulin proteins form antibodies. Most chemical messengers or hormones are steroid in nature. Thrombin is a biocatalyst involved in the clotting of blood.
Explanation:-
Answer: Option C. -> Sphingolipids:
C
Sphingosine (2-amino-4-octadecene-1,3-diol)isan 18-carbon amino alcohol with an unsaturated hydrocarbon chain, which forms a primary part of sphingolipids. The derivatives of Sphingosine and fatty acids are what forms Sphingolipids.
Explanation:-
Answer: Option A. -> Vitamin-D:
A
Vitamin D helps to absorb calcium and phosphorus, and it's deficiency in the body, causes rickets.
Vitamin A is needed for healthy bones, eyes, skin and also acts as an anti-oxidant. It's deficiency in the body causes night blindness and hyperkeratosis.
Vitamin K is required in the body for blood clotting cascade and for bone metabolism. It's deficiency causes bleeding diathesis.
Vitamin E acts as an anti-oxidant and is also vital for smooth muscle development. It's deficiency in infants, leads to hemolytic anemia.
Explanation:-
Answer: Option D. -> Valine:
D
Amino acids that dissolve in water are hydrophilic amino acid, that is the R- group in these amino acids can interact with water, in contrast with the hydrophobic amino acids that do not interact with water and thereby do not dissolve in it. Of the amino acids listed here valine, with a (CH3−CH−CH3) R-group is the hydrophobic amino acid.
Explanation:-
Answer: Option C. -> Thermal Denaturation:
C
Application of heat alters the secondary, tertiary and quaternary structure of proteins. This phenomenon is called thermal denaturation. It is used as a means to study protein structures, folding and polymerisation.
Explanation:-
Answer: Option D. -> There is no internal transfer of charge in amino acids:
D
Amino acids contain an alkali amine group and an acidic carboxyl group, both of which exists in the ionic form in solutions. The caboxyl group loses a proton, which is taken up by the amine group. Therefore amino acids contain both positive and negative ion, forming a zwitter ion. Diffraction studies reveal that amino acids also exist as zwitter ions in the solid state.